Influenza Diagnostics Market to Cross US$ 2.5 Bn Valuation by 2030; Increased Need for Influenza Testing Amid Covid-19 Pandemic Shoring Up Demand
Influenza, or flu, is a respiratory infection caused by viruses. According to the WHO, the average global burden of seasonal influenza is in the order of 600 million cases, with 3 million cases of severe illness and 250,000 - 500,000 deaths per year. Diagnostic tests available for influenza include rapid reverse transcription-polymerase chain reaction (RT-PCR), viral culture, immunofluorescence assays, serology, antigen testing, and rapid molecular assays.
However, commercial rapid influenza diagnostic tests are mostly used for influenza detection, owing to their ability to detect influenza viruses within 15 minutes, with low to moderate sensitivity and high specificity. Since the past few years, RT-PCR tests and nucleic acid amplification technique have been widely developed in clinical microbiology labs for routine influenza infection diagnostics. This factor will boost the growth of the influenza diagnostics market during the forecast period.
Get Sample Copy of this Report @ https://www.persistencemarketresearch.com/samples/13246
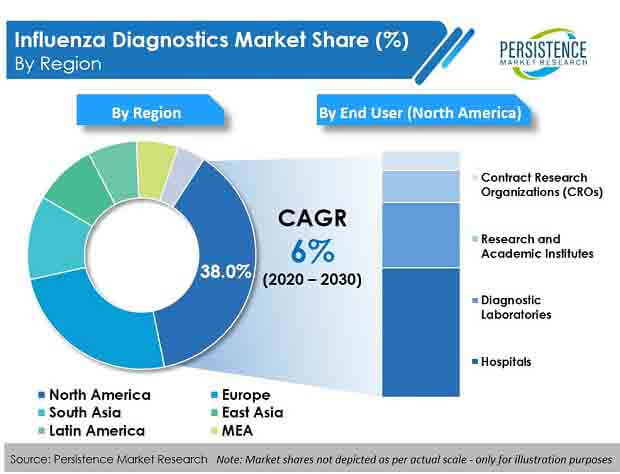
According to a latest report published by PMR, the global influenza diagnostics market is projected to account for over US$ 2.5 Bn by 2030, in terms of value. The report further projects that, the influenza diagnostics market will expand at a CAGR of 6% through 2030.
Companies
Request for Methodology@ https://www.persistencemarketresearch.com/methodology/13246
Key Takeaways of Influenza Diagnostics Market Study
“Availability of commercial rapid influenza diagnostic tests, and seasonal flu outbreaks and their complications every year, are expected to propel the growth of the global influenza diagnostics market,” says a PMR analyst.
Access Full Report@ https://www.persistencemarketresearch.com/checkout/13246
Acquisitions & Partnerships - Key Strategies amongst Market Players
Key players in the influenza diagnostics market are focussed on improving their product portfolios though the launch of new products. For instance, In April 2017, F. Hoffmann-La Roche Ltd launched the cobas Liat PCR System with four assays for respiratory disease diagnosis. In February 2019, GenMark Diagnostics, Inc launched a multiplex serological assay to identify avian influenza subtypes. Various players in the influenza diagnostics market are focusing on growth strategies such as acquisitions and partnerships. For example, in September 2016, Danaher Corporation announced the acquisition of Cepheid at approximately US$ 53 per share in cash.
What Does the Report Cover?
Persistence Market Research offers a unique perspective and actionable insights on the influenza diagnostics market in its latest study, presenting historical demand assessment of 2015–2019 and projections for 2020–2030, on the basis of test (molecular diagnostic tests, traditional diagnostic tests), type of flu (type A flu, type B flu, type C flu), and end user (contract research organizations (CROs), research & academic institutes, hospitals, and diagnostic laboratories), across seven key regions.
About us: Persistence Market Research
Contact us:
Persistence Market Research
Address – 305 Broadway, 7th Floor, New York City,
NY 10007 United States
U.S. Ph. – +1-646-568-7751
USA-Canada Toll-free – +1 800-961-0353
About Us: Persistence Market Research (PMR), as a 3rd-party research organization, does operate through an exclusive amalgamation of market research and data analytics for helping businesses ride high, irrespective of the turbulence faced on the account o
Jun 28, 2022